It is of great interest to identify phosphorylated amino acid residues in proteins. Phosphorylation occurs in the serine, threonine and proline of the protein, and the labeled phosphoric acid amino acid can be conveniently identified by partial HCl hydrolysis followed by two-dimensional thin layer electrophoresis.
experimental method
- Acid hydrolysis
Phosphorylated protein | |
Indian ink HCl phosphoric acid amino acid mixture running buffer ninhydrin | |
PVDF membrane oven centrifuge centrifuge tube cellulose thin layer chromatography filter paper thin layer electrophoresis | |
| Experimental procedure | 1. Radiolabeled phosphorylated protein was prepared for electrophoresis on an SDS-polyacrylamide gel. Transfer the electricity to the PVDF membrane and wash the membrane several times with water. Do not let the membrane dry. 2. Stain the film with 30~50 ml Indian ink staining solution for 5~10 min and gently shake until the strip appears. It is also possible to carry out autoradiography by encapsulating the film with a plastic film after the radioactive or phosphorescent position mark is made. 3. Cut the membrane containing the desired strip with a clean blade, rewet the membrane with methanol for 1 min, and wet with more than 0.5 ml of water. Place in a microcentrifuge tube with a screw cap. 4. Add enough 6 mol/l HCl to submerge the membrane, tighten the tube cap, and incubate in a 110 °C oven for 60 min. 5. Naturally cool, centrifuge at high speed for 2 min, transfer the hydrolyzate from the liquid phase to a new microcentrifuge tube and dry in vacuum for 2 h. 6. Add 6~10 μl of water, shake vigorously on a vortex mixer to dissolve the sample, and centrifuge at high speed for 5 min. 7. Take 25% to 50% of the sample to the sample well of a 20 cm × 20 cm × 100 μm cellulose thin-layer chromatography plate. Add a small portion, add 0.25~0.5 μl to the next point, and blow the sample point with compressed air sent through a Pasteur pipette with cotton between each point. 8. Take 1 μl of a non-radioactive phosphate amino acid standard mixture (containing phosphoserine, phosphothreonine and phosphotyrosine) on each sample, add 0.25 to 0.5 μl each time. 9. In a large glass tray or plastic box, soak a large absorbent paper (with 4 holes) with pH 1.9 running buffer and slowly pour off the excess buffer. Transfer the blotting paper over the loaded TLC plate so that the four loading points on the chromatographic plate are in the center of the four holes of the absorbent paper, and gently press the absorbent paper to wet the fibronectin. And the sample, after the chromatographic plate is evenly wetted, remove the absorbent paper. 10. Place the TLC plate in the electrophoresis unit and lap it on the left and right 0.5 cm edges of the plate with Whatman 3MM paper. If there are air bubbles, flatten it out. Cover and start electrophoresis. When the HTLE 7 000 electrophoresis instrument was lapped with double-layered Whatman 3MM paper, the thin plate containing 4 samples was electrophoresed at 1.5 KV for 20 min. 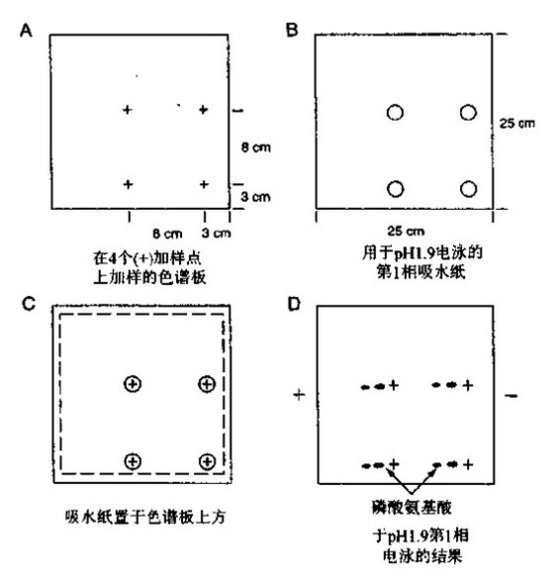 Figure 1 First-line electrophoresis separation of phosphoric acid amino acids at pH 1.9 11. After electrophoresis, remove the thin plate and blow dry quickly with a hair dryer for 20 min (without heating). 12. Dampen the small absorbent paper with pH 3.5 running buffer and uniformly wet the TLC as described in step 10, taking care to avoid placing the blotting paper on top of the sample. 13. Remove the small absorbent paper and turn the chromatographic plate back 90 times . Perform the second-direction electrophoresis. If using an HTLE 7 000 electrophoresis apparatus, electrophoresis was carried out for 16 min at 1.3 KV using an electrode buffer of pH 3.5. 14. After electrophoresis, remove the laminate and dry it in the oven at 50~80 °C for 20~30 min until it is completely dry. Spray 0.25% ninhydrin acetone solution and heat for 5~10 min to make the phosphoric acid amino acid standard trace. 15. Add a radioactive or phosphorescent locating marker to the TLC and auto-develop at -70 °C using an intensifying screen from overnight to approximately 10 days. 16. After auto-development, mark the position of the standard phosphoric acid amino acid on the transparent transparencies with the positioning marks and staining traces, and retain the chromatographic plate. The radioactive amino acid amino acid can be identified by comparing the slide and the photosensitive sheet. 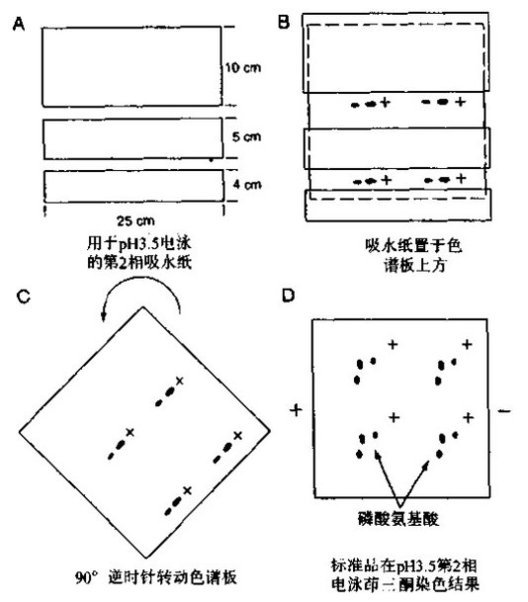 Figure 2 Amino acid separation of phosphoamino acid at pH 3.5 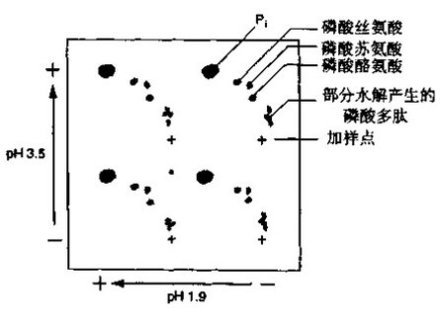 |
Vitamin is a kind of trace organic matter that human and animals must obtain from food in order to maintain normal physiological function. It plays an important role in the process of human growth, metabolism and development. Vitamins neither participate in the formation of human cells nor provide energy for the human body.
Vitamins are a class of organic compounds necessary to maintain good health. This kind of substance is neither the raw material of body tissue nor the source of energy in the body, but a kind of regulatory substance, which plays an important role in material metabolism. This kind of substance cannot be synthesized or the synthetic amount is insufficient in the body, so although the demand is small, it must be often supplied by food.
Vitamins are also called vitamins. Generally speaking, they are life sustaining substances. They are a kind of organic substances necessary to maintain human life activities, and they are also important active substances to maintain human health. Vitamin content in the body is small, but indispensable. Although the chemical structures and properties of various vitamins are different, they have the following common points:
â‘ Vitamins exist in food in the form of provitamin;
â‘¡ Vitamin is not a component of body tissues and cells, and it will not produce energy. Its role is mainly to participate in the regulation of body metabolism;
â‘¢ Most vitamins cannot be synthesized by the body or the synthetic amount is insufficient to meet the needs of the body, and must be often obtained from food;
â‘£ The human body's demand for vitamins is very small. The daily demand is often calculated in mg or microgram, but once it is deficient, it will lead to corresponding vitamin deficiency and damage human health.
Different from carbohydrate, fat and protein, vitamins only account for a small proportion in natural food, but they are necessary for the human body. Some vitamins such as B6 and K can be synthesized by bacteria in the intestinal tract of animals, and the synthetic amount can meet the needs of animals. Animal cells can convert tryptophan into niacin (a B vitamin), but the production does not meet the needs; Vitamin C can be synthesized by other animals except primates and guinea pigs. Plants and most microorganisms can synthesize vitamins themselves without having to be supplied in vitro. Many vitamins are components of cofactors or coenzymes.
Vitamins are some small amounts of organic compounds necessary for human and animal nutrition and growth. They play a very important role in the metabolism, growth, development and health of the body. If you lack a certain vitamin for a long time, it will cause physiological dysfunction and some diseases. It is usually obtained from food. At this stage, there are dozens of found, such as vitamin A, vitamin B, vitamin C, etc.
Vitamins are essential organic compounds in human metabolism. The human body is like an extremely complex chemical plant, constantly carrying out various biochemical reactions. The reaction is closely related to the catalysis of enzymes. Coenzyme must be involved in enzyme activity. Many vitamins are known to be coenzymes of enzymes or components of coenzymes. Therefore, vitamins are important substances to maintain and regulate the normal metabolism of the body. It can be considered that the best vitamins exist in human tissues in the form of "bioactive substances".
50% Vitamin Injection,50% Vitamin Injection for Vet,Vitamin 50% Injection,100ML Vitamin Injection
Shandong Unovet Pharmaceutical Co.,Ltd. , https://www.unovetcn.com