Release date: 2018-03-13
Recently, Xeris Pharmaceuticals, a biotechnology company focused on ready-to-use injection preparations, announced that it has signed a $45 million loan bond agreement with Silicon Valley Bank and Oxford Finance, and completed a $10 million financing in Series C. The funds obtained will be used to support the preparation and commercialization of new drugs for the preparation of “ready-to-use glucagon emergency injection pens†to rescue severe hypoglycemia. This round of C financing was led by Merieux Developpement, and Wild Basin Investments and Xeris management participated in the investment.

Xeris specializes in the development and commercialization of ready-to-use liquid-stable injections with its proprietary two new formulation technology platforms, XeriSolTM and XeriJectTM, for high-concentration, non-aqueous peptides, proteins, antibodies and small molecule drugs. A ready-to-use subcutaneous and intradermal administration dosage form based on prefilled syringes, autoinjectors, multi-dose pens and infusion pumps has been developed. This ready-to-use injection helps eliminate pre-injection reconstitution, achieves long-term room temperature stability, significantly reduces the amount of each injection, and eliminates the complex need for intravenous (IV) injection.
Its leading product, the ready-to-use glucagon emergency injection pen, has obtained key phase 3 trial data for the rescue of hyperinsulinemia-associated hypoglycemia and has been qualified as an FDA orphan drug. Other target indications for research include: post-bariatric hypoglycemia after laparoscopic gastric bypass, congenital hyperinsulinemia, autoglycemia associated with hypoglycemia, exercise-induced hypoglycemia, and Double hormone auto-closed artificial pancreas system related to hypoglycemia complications.
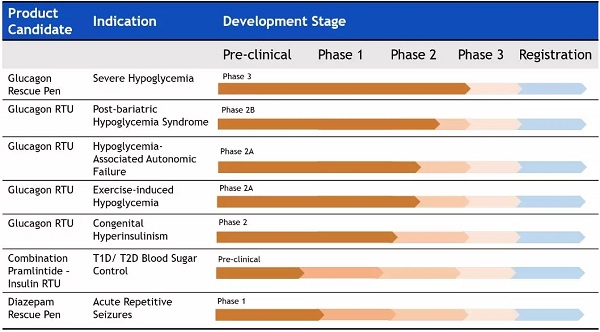
â–²Xeris' R&D pipeline (Source: Xeris official website)
“Hyperglycemia is a fatal complication after bariatric surgery,†said Mary-Elizabeth Patti, associate professor of medicine at the Joslin Diabetes Center at Harvard Medical School. “There is an urgent need to improve the recovery of this type of postoperative severe hypoglycemia. Ways to keep patients healthy, get the best nutrition and improve safety. Xeris' high solution stability glucagon ready-to-use injections offer a new convenience option to help healthcare professionals and patients prevent serious Hypoglycemia."
Glucagon is the gold standard for the rescue of severe hypoglycemia events. However, many people are currently unable to carry glucagon at any time. Part of the reason is due to the limitations of the current selection of glucagon preparations, which are almost all powders. Inconvenient use requires a lengthy, complicated, time-consuming multi-step reconstitution solution and a dose calibration process, which presents management risks. Xeris' stable glucagon ready-to-use injections will bring new breakthroughs.
Reference materials:
[1] Xeris Raises $55M for Glucagon Rescue Pen
[2] Xeris Pharmaceuticals official website
Source: WuXi PharmaTech
COVID-19 Antigen Rapid Test-nasal Swab
Covid-19 Antigen Rapid Test-Nasal Swab,Covid-19 Antigen Rapid Test With Swab,Covid 19 Antigen Pre-Nasal Test Cassette,Medical Detection Covid-19 Rapid Test Kit
Weihai Kangzhou Biotechnology Engineering Co.,Ltd , https://www.weihaikangzhou.com