Medical Network March 11th, March 8th, Sichuan Kelun Pharmaceutical announced the company's products "Fluconazole tablets (50mg)" and "Acyclovir tablets (0.1g, 0.2g, 0.4g)", Through the evaluation of generic consistency.

Fluconazole tablets
Fluconazole is a triazole antifungal drug widely used in the treatment and prevention of fungal infections. It has the advantages of rapid oral absorption, high bioavailability, long half-life, clear efficacy and good safety. At present, fluconazole tablets have entered the national basic drug list and the national medical insurance catalog.
According to the statistics of Mine Net, the sales in China in 2017 was approximately RMB 110 million.
The company's announcement shows that Cologne is the first domestic company to pass the consistency evaluation of the variety. Up to now, the company has invested R&D expenses of approximately RMB 3.89 million in the fluconazole consistency evaluation project.
According to the data of the State Food and Drug Administration, there are 24 domestically approved fluconazole tablets and 17 production enterprises in China. According to the statistics of Yaozhi.com, four pharmaceutical companies including Sichuan Kelun Pharmaceutical, Yuekang Pharmaceutical, Yangzijiang Pharmaceutical and Shijiazhuang Pharmaceutical have accepted the conformity evaluation and have completed the BE test. In time, Sichuan Kelun Pharmaceutical was the first to comment.
Report on the consistency evaluation of fluconazole tablets:
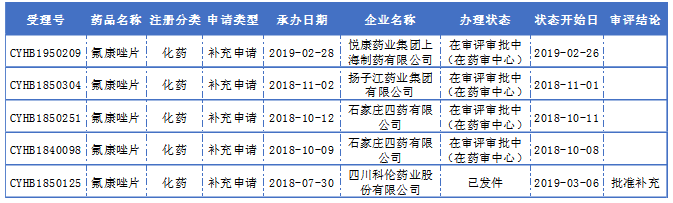
(Source: Yaozhi Network)
Acyclovir tablets
Acyclovir belongs to purine nucleoside analogues and is mainly used for infectious diseases caused by various viruses such as herpes simplex virus, varicella zoster virus and cytomegalovirus. It is a widely used broad-spectrum antiviral drug in clinical practice. It has the characteristics of high efficiency, low toxicity and high selectivity to inflammatory cells.
At present, acyclovir tablets have entered the national drug list and the national medical insurance catalog.
According to the statistics of the intranet, China's sales in 2017 were about 114 million yuan. According to the company's announcement, as of now, Cologne has invested R&D costs of approximately 7.92 million yuan in the acyclovir tablet consistency evaluation project.
According to the data of the State Food and Drug Administration, there are 100 domestic approvals for acyclovir tablets in China, involving 85 production enterprises. According to the statistics of Yaozhi.com, only Shandong Qidu and Sichuan Kelun Pharmaceuticals have submitted a consistent evaluation.
Acyclovir tablets (0.2g) were the first to be evaluated by Shandong Qidu Pharmaceutical Co., Ltd., and Sichuan Kelun was the second company to evaluate the product. However, acyclovir tablets (0.1g, 0.4g) two specifications, Sichuan Kelun is the first company to comment.
The consistency of acyclovir tablets:
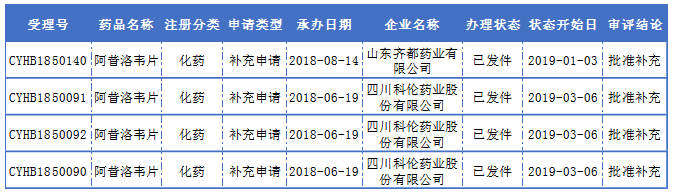
(Source: Yaozhi Network)
Kelun Pharmaceutical, 12 product reviews
At present, it is still the peak period for the consistency assessment of major pharmaceutical companies. Despite the cancellation of the deadline, but in 2019, the enthusiasm of pharmaceutical companies to report consistency evaluation is not reduced.
According to statistics from Guotai Junan, this year's generic drug consistency application was hot, and the number of single-month acceptances surged from 111 in December last year to 209 in January this year, setting a new high. From the completion rate, the company is also sprinting. In December last year, the number of varieties passed in a single month reached 32, also hitting a new high.
Among them, amlodipine besylate tablets are the most competitive, and 6 companies have passed the consistency evaluation. There are still 30 companies in the declaration. The tenofovir disoproxil fumarate tablets have been approved by 5 companies, and the amoxicillin capsules have been approved by 4 companies, with a total of 29 reviews.
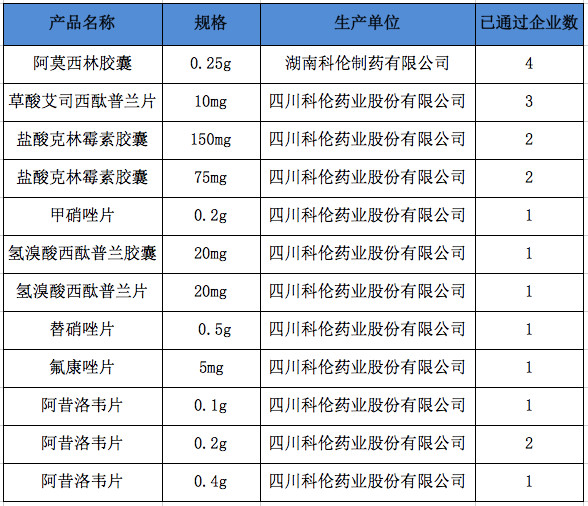
It is reported that there are currently 44 acceptance reports for the consistency evaluation of Kelun Pharmaceutical, including 23 from Sichuan Kelun Pharmaceutical, 17 from Hunan Kelun Pharmaceutical, 3 from Hubei Kelun Pharmaceutical, and 1 from Heilongjiang Pharmaceutical.
According to cypress blue statistics, up to now, 198 drugs have passed the consistency evaluation, involving a total of 143 product specifications. Cologne has 12 articles that pass the consistency evaluation, and 7 of them are currently passed the consistency evaluation. Details are as follows:
Split Drape,U Split Drape,U Shape Drape,Surgical Split Drape
Xinxiang Huaxi Sanitary Materials Co., Ltd. , https://www.huaximedical.com