Caring for women's health! The approval of new drugs is difficult to say
May 31, 2018 Source: WuXi PharmaTech
Window._bd_share_config={ "common":{ "bdSnsKey":{ },"bdText":"","bdMini":"2","bdMiniList":false,"bdPic":"","bdStyle":" 0","bdSize":"16"},"share":{ }};with(document)0[(getElementsByTagName('head')[0]||body).appendChild(createElement('script')) .src='http://bdimg.share.baidu.com/static/api/js/share.js?v=89860593.js?cdnversion='+~(-new Date()/36e5)];Today, TherapeuticsMD, a company dedicated to the development of innovative women's health products, announced that the US FDA has approved its new drug, Imvexxy (estradiol vaginal insert), for the treatment of moderate to severe sexual intercourse pain (vaginal pain associated with sexual activity). Vulvar and vaginal atrophy (VVA) symptoms due to menopause. It is worth mentioning that Imvexxy is the only product in its treatment category that offers doses of 4 micrograms and 10 micrograms, and 4 micrograms is the lowest approved dose of vaginal estradiol currently available.

VVA is part of the menopausal genitourinary syndrome (GSM). Genital symptoms are dry, burning and irritating. Sexual symptoms include reduced lubrication, discomfort and pain. Urinary symptoms include urgency, difficulty urinating and recurrent urinary tract. infection. VVA is a chronic progressive condition that can cause discomfort and gradually worsens if not treated promptly. VVA affects approximately 32 million postmenopausal women in the United States, and only 7% (2.3 million) of them are receiving prescription treatment. 81% of women do not know that VVA is a treatable disease, and they have unmet medical needs.
Imvexxy from TherapeuticsMD re-estrogenizes the intravaginal and surrounding tissues. Its unique formula ensures complete dissolution of the drug so patients can use it at any time of the day. Imvexxy usually needs to be used for 2 weeks per day, and then can be used twice a week. Nine of the 10 patients who participated in the clinical trial reported that the drug was “easy to useâ€.
The Imvexxy FDA approval was based on a randomized, double-blind, placebo-controlled Phase 3 study. The study evaluated the efficacy and safety of Imvexxy (4 micrograms and 10 micrograms) from baseline to week 12 compared to placebo. Studies have shown that two doses of Imvexxy have been able to alleviate moderate to severe sexual intercourse pain due to menopause at week 2. A subgroup of the REJOICE trial also evaluated the pharmacokinetics of Imvexxy (4 micrograms and 10 micrograms) and placebo. The results showed that the average concentration of estradiol and estrone was still within the average postmenopausal range at both doses. In terms of safety, no significant clinically significant adverse events (AEs) were observed between the treatment and placebo groups.
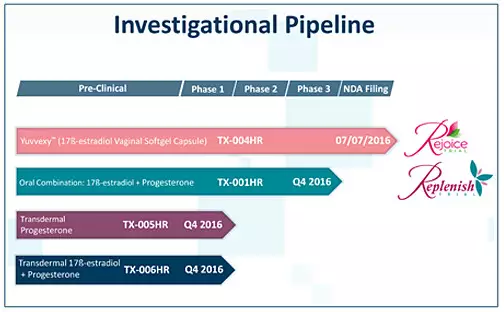
â–²TherapeuticsMD has a variety of research products (Source: TherapeuticsMD official website)
“Imvexxy is a biologically identical vaginal estrogen product that provides a fraction of the average estrogen dose of many existing products on the market,†said Dr. Brian Bernick, Chief Clinical Officer of TherapeuticsMD. “Imvexxy is the only design specifically designed. It is a product without applicator. It dissolves completely, does not cause confusion, or requires extra cleaning, and the patient can use it at any time of the day. It allows women to quickly return to normal daily activities. Studies have shown that In patients who used Imvexxy, the systemic absorption of estradiol remained within the postmenopausal range."
“We are very excited to bring Imvexxy to market. It is TherapeuticsMD's first FDA-approved drug, and we are working hard to become a good women's health company,†said Robert Finizio, CEO of TherapeuticsMD. “Imvexxy reflects our long-term Our corporate mission and commitment to addressing women's health needs are based on medical needs, efficacy, safety, simplicity and affordability. Imvexxy will be available at the same price as other products that have been on the market for 10 to 30 years. To ensure that patients get Imvexxy at a reasonable price. Through these, TherapeuticsMD is doing the right thing for women."
Reference materials:
[1] TherapeuticsMD Announces FDA Approval of TX-004HR: IMVEXXYTM (estradiol vaginal inserts), the Lowest Dose Vaginal Estrogen Product Approved for the Treatment of Moderate to Severe Dyspareunia, a Symptom of VVA, due to Menopause
[2] On Second Shot TherapeuticsMD Snags FDA Approval for VVA Medication
Vegetarians Size 00 Empty Capsule
Vegetarians Size 00 Empty Capsule,Gelatin Empty Pills Capsules Vegan,Colorful Vegan Empty Pills Capsules,Size 00 Empty Pills Capsules Vegan
Ningbo Jiangnan Capsule Co., Ltd. , https://www.ningbocapsule.com