Eisai publishes independent imaging review of levastatin hepatocellular carcinoma
January 23, 2018 Source: Sina Pharmaceutical
Window._bd_share_config={ "common":{ "bdSnsKey":{ },"bdText":"","bdMini":"2","bdMiniList":false,"bdPic":"","bdStyle":" 0","bdSize":"16"},"share":{ }};with(document)0[(getElementsByTagName('head')[0]||body).appendChild(createElement('script')) .src='http://bdimg.share.baidu.com/static/api/js/share.js?v=89860593.js?cdnversion='+~(-new Date()/36e5)];On January 22, the company's Tokyo-based Eisai Pharmaceutical announced that the company's anticancer drug leventinib mesylate (trade name: Lenvima® / Kisplyx®) was compared with sorafenib for unresectable liver. The clinical phase 3 trial of first-line treatment of cell carcinoma based on independent imaging analysis has been published in the 2018 American Society of Clinical Oncology Gastrointestinal Cancer Symposium (ASCO-GI).

The study published an exploratory analysis of blind progression independent imaging review (IIR) for progression-free survival (PFS), tumor progression time (TTP), and objective response rate (ORR) for the secondary phase of the three-phase REFLECT study.
The IIR review is based on the solid tumor efficacy evaluation standard RECIST1.1 (which uses traditional methods for assessing tumor diameter changes) and the revised RECIST evaluation protocol (mRECIST), in which mRECIST examines tumor necrosis in addition to the 1.1 version of the standard. area. The results of the IIR review confirmed the conclusion that lenvatinib has the clinical effect of prolonging PFS, TTP and increasing ORR (lenvatinib can better inhibit tumor volume) compared with sorafenib. The REFLECT study blinded IIR results supported tumor imaging findings observed by clinical researchers. The analysis results are as follows:
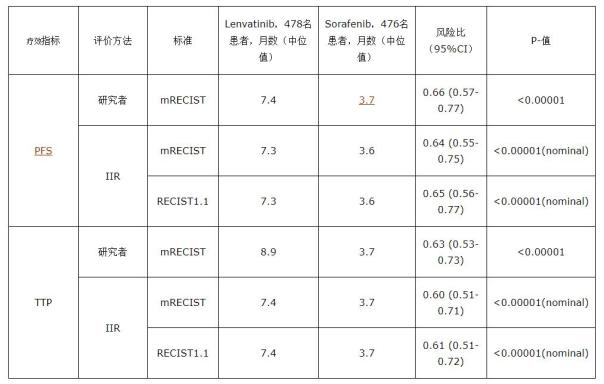
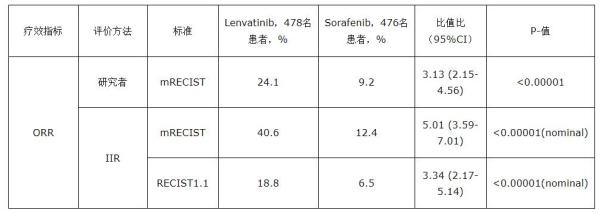
In this phase 3 study, the most common adverse events observed in the lenvatinib group were hypertension, diarrhea, loss of appetite, weight loss, and fatigue, which are consistent with the known safety risks of lenvatinib.
Liver cancer is the second largest cancer causing cancer-related deaths, and it is estimated that 750,000 people worldwide die of liver cancer each year. In addition, there are 780,000 newly diagnosed cases each year, of which about 80% occur in Asia, including Japan and China. Hepatocellular carcinoma accounts for 85%-90% of the total incidence of liver cancer. Early hepatocellular carcinoma can be treated by a variety of methods, including surgery, radiofrequency ablation, ethanol injection, and chemoembolization. However, the treatment options for difficult-to-clear hepatocellular carcinoma are limited and the prognosis is poor, which means that this is a highly unmet medical field for treatment needs.
In June 2017, Eisai submitted a supplemental listing application for lenvatinib for hepatocellular carcinoma treatment in Japan. The registration dates for the United States, Europe, China and Taiwan are July 2017, July 2017, and October 2017. December 2017. Eisai remains committed to scientific evidence for maximizing the value of lenvatinib, as lenvatinib has the potential to further meet the diverse needs of cancer patients, their families and health care providers, and to benefit them. (Sina Pharmaceutical Compilation / David)
Article reference source: http://
Erythritol For Jellies,Organic Erythritol,Erythritol Of Xylitol,Erythritol Liquid Sweetener
Ningxia Eppen Biotec CO.,LTD , https://www.nxeppen.com